AI-powered endpoints and analytics
Qureight has developed a suite of deep learning–based quantitative imaging models that generate compartment-specific biomarkers directly from standard-of-care inspiratory CT scans. These endpoints are purpose-built for interventional clinical trials and are linked to treatment response, disease progression, and long-term clinical outcomes.
“CT imaging contains an extraordinary amount of biological information, but historically we have reduced it to subjective impressions or crude categorical scores. Our AI models transform routine CT scans into precise quantitative biomarkers, allowing clinical trials to measure disease biology directly rather than relying solely on downstream functional surrogates.”
Dr Simon Walsh
Chief Scientific Officer
Our models are fully CT vendor-agnostic, validated across varying CT acquisition protocols, and require no respiratory gating or phantom calibration. Powered by full 3D volumetric deep convolutional neural networks, they deliver robust, reproducible quantification at scale. In peer-reviewed publications, our platform has demonstrated a 98.4% successful CT analysis rate.
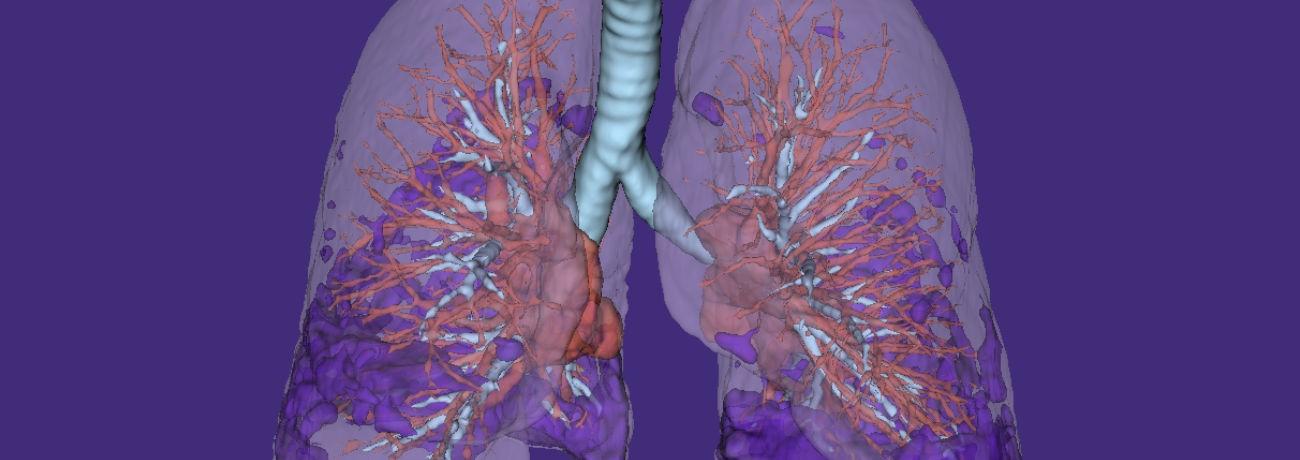
Each model isolates a biologically distinct lung compartment, enabling precise detection of compartment-level treatment effects.
Fibr8™ quantifies lung fibrosis.
Glass8™ quantifies inflammatory (ground glass) change.
Air8™ quantifies airway volume.
Vascul8™ quantifies pulmonary vascular volume.
Lung8™ measures whole-lung and lobar volumes.
By measuring these compartments independently rather than relying on composite or indirect markers, Qureight enhances sensitivity to disease change, enables clearer interpretation of therapeutic impact, and provides compartment-specific mechanistic insight into drug effect
Beyond endpoint generation, Qureight provides real-time enrolment analytics
Imaging-derived biomarkers are analysed as patients are enrolled, allowing sponsors to monitor disease severity distributions and benchmark enrolled cohorts against predefined trial criteria and reference populations. This enables early detection of enrolment drift, supports proactive course correction during recruitment, and reduces late-stage trial failure risk by protecting cohort integrity before database lock.
-
Elucidation of a drug mechanism for a biotech partner
Endeavor BioMedicines conducted a randomised, double-blind, placebo-controlled Phase 2a trial of ENV-101, a novel Hedgehog (Hh) pathway inhibitor in IPF, with a hypothesised vascular mechanism that had not previously been demonstrated in patients. Qureight applied Vascul8™ to baseline and end-of-study CT scans, demonstrating a statistically significant reduction in pulmonary vascular volume over 12 weeks in the active treatment arm compared with placebo (p=0.0007). These findings provided in vivo evidence supporting the drug’s proposed mechanism and were presented at the American Thoracic Society in 2025, informing progression into Phase 2b development.
-
Analysis of combined imaging and proteomic data with a biopharma partner
Our partner AstraZeneca had a specific interest in proteomic biomarkers in lung fibrosis as markers of baseline progression, which could be used to enrich cohorts for clinical trials. Working with them on a large clinical IPF dataset (PROFILE), Qureight ran multiple deep learning imaging models on baseline data and combined those outputs with proteomic biomarkers of interest. Using Fibr8TM, we showed that deep learning measures of fibrosis protein biomarkers of epithelial injury and collagen turnover (CYFRA21-1, SPD, and ProC6) predicted mortality in IPF, independently of physiological biomarkers such as FVC. These data were presented at the American Thoracic Society meeting in 2025.
Find out how we can
suport your clinical trial