“At Qureight, we didn’t just build an imaging CRO—we reimagined it from the ground up. Every tool, every workflow, every process was designed to tackle the real pain points of clinical trials: the delays, the inefficiencies, and the frustration of unusable data. We’ve turned slow study start-ups into rapid launches, complex site initiations into seamless onboarding, and poor data quality into precision insights. Welcome to the new standard for clinical trials.”
Dr Steven Bishop
Chief Medical Officer
Key advantages of our CRO services
1. Efficient and effective study management
Our services are designed for efficient study start-up, including a cloud-hosted, globally available platform and automated validation for improved adherence to the Imaging Charter.
2. Site centric / friendly
Our tiered remote site qualification process is built for fast site activation and protection of First Patient In (FPI) timelines. We support a wide range of real-world CT scanners and protocols. There is no need for complex or time-consuming image acquisition, phantom scans or respiratory gating.
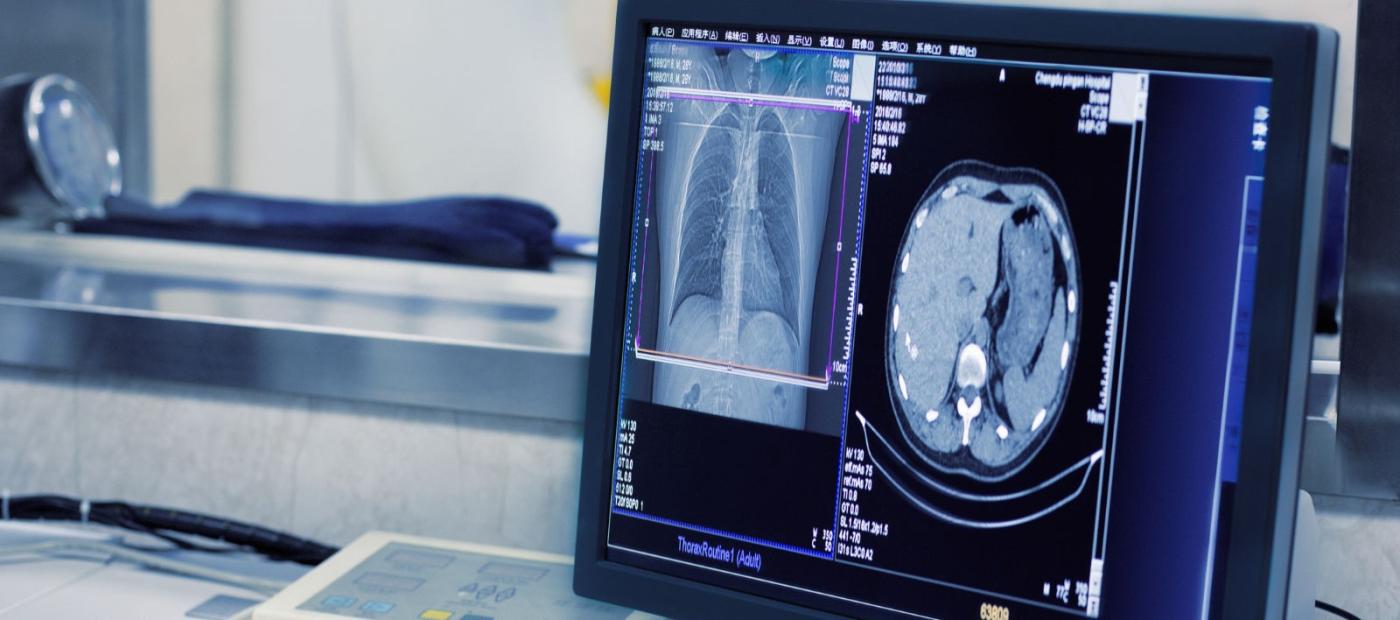
3. Fast, accurate central read turnaround
We provide expert central reader teams comprising named radiology key opinion leaders with extensive clinical trial experience to improve consistency. Our read forms are co-designed by our experts to help you maximise utility and consistency. We guarantee a 72-hour turnaround time for all eligibility reads.
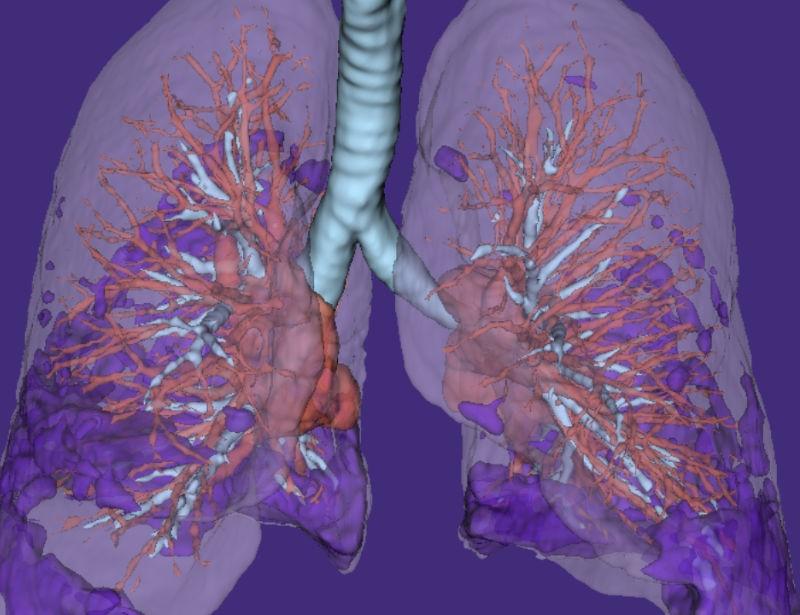
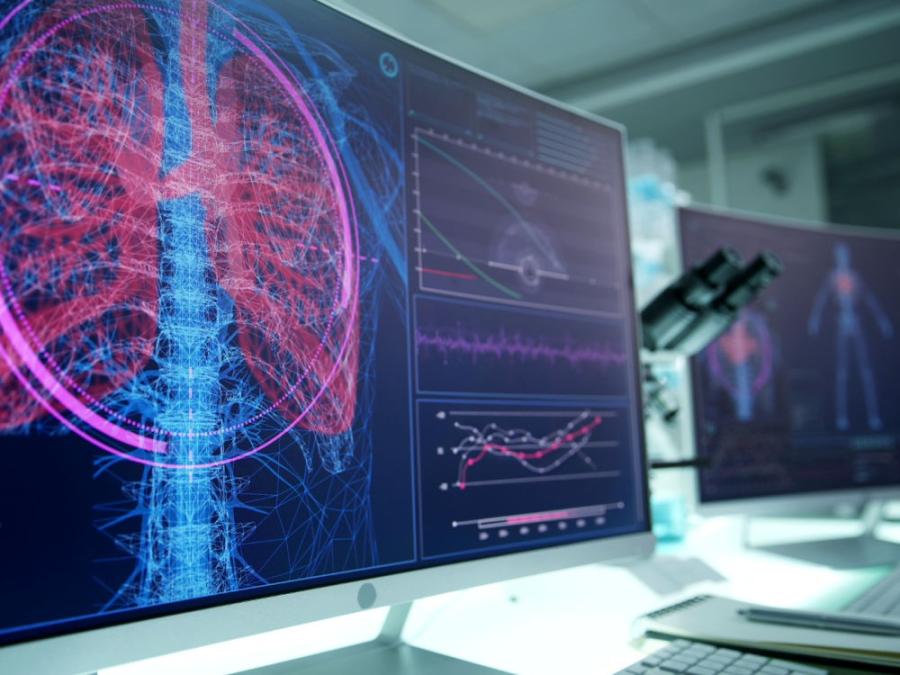
4. Integrated AI insights and endpoints
Qureight's AI-powered endpoints and analytics are integrated into our platform. These support cohort enrichment strategies and automated reporting of disease severity across all enrolled patients.
5. Built for compliance
The Qureight platform provides secure image transfer, global data privacy compliance, built-in quality controls and full auditability. It’s compliant with GCP, GAMP 4/5, EMA CSV, 21 CFR Part 11, ISO 27001, ISO 13485, GDPR, and HIPAA.
6. Backed by world-leading expertise
We provide access to globally recognised key opinion leaders in lung and heart disease, including in-house expertise, our disease-specific Scientific Advisory Boards and advisor KOLs.